So what has research done for the horse breeder lately? |
||||
|
||||
Dr. Keith Betteridge |
Department of Biomedical Science Ontario Veterinary College, University of Guelph |
|||
|
||||
Since the 1980s, reproductive management of the mare without ultrasonography has become unthinkable; assessments of ovarian function, embryonic development, and uterine physiology and pathology all depend upon it. While marvelling at the images on echograph screens, however, spare a thought for the research that went into working out what those images mean and where and when to look for them. Whereas rectal palpation is subjective, ultrasonography allows measurements to the millimeter, sometimes images in colour and/or three dimensions, and reveals subtle changes that help the clinician predict when ovulation is likely to occur and to time insemination accordingly, thereby maximizing the chances of fertilization.
To hasten ovulation the mare may be injected with human chorionic gonadotrophin (hCG), a hormone prepared from the urine of pregnant women. The use of hormones in reproduction became more rational after research during the 1960s and 1970s made it possible to measure their concentrations in blood during the estrous cycle and early pregnancy.
Gonadotrophin releasing hormone (GnRH), which is released from the hypothalamus of the brain and stimulates the pituitary gland to release luteinizing hormone, is an essential link in the pathway to ovulation. The biological activity of GnRH was demonstrated in the1960s; identifying its structure resulted in two Nobel Prizes for medicine in 1977. Structural modifications of GnRH have uses as varied as the treatment of prostate cancer in men, preparation for in-vitro fertilization (IVF) in women, and the induction of ovulation in mares – an example of the truism that one never knows where products of use to horse reproduction will come from.
That truism also applies to the prostaglandin often used to bring mares into heat. If, in the late 1960s, a researcher had applied for funding to work on a coral-like organism to help horse reproduction, he or she would probably have been laughed out of court. Yet it was the abundance of prostaglandins in the sea-whip that enabled the Upjohn Company to produce the compounds more cheaply and to make their use widespread. One never knows where products of use to horse reproduction will come from!
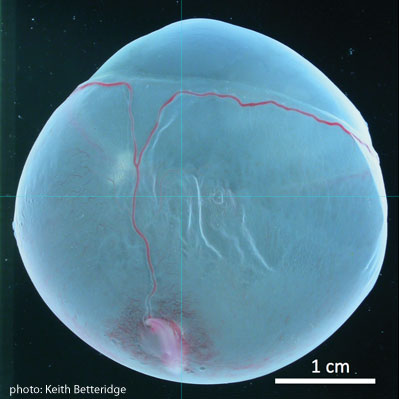
A complementary relationship between rectal palpation and ultrasonography shows up in relation to pregnancy diagnosis. It was sensitive fingers that first revealed that the equine conceptus (the embryo and its fluids and membranes; see figure) moves around in the uterus; ultrasonography substantiated that fact in the early 1980s and has enabled us to “see” conceptus movement until about Day 16 of pregnancy when it becomes fixed where the placenta will form. Some 17% of pregnancies detected by ultrasonography at about Day 15 will not result in a foal, and most (60%) of those losses will be incurred before Day 35.[3] Researchers at Guelph are paying special attention to that period, studying how the conceptus attaches to the endometrium (lining of the uterus).
Techniques for studying early pregnancy in horses have been revolutionized over the past three decades through efforts to produce and manipulate embryos in the laboratory (in vitro) and the techniques of molecular biology – genomics and proteomics – that have culminated in the sequencing of the equine genome.[4]
The production of embryos in vitro requires detailed knowledge of sperm and ova and how they interact at fertilization. Research into the stallion and his semen dates to the earliest days of artificial insemination[5] and remains a major subject. The direct benefits to the horse breeder of being able to ship cooled or frozen semen over long distances and time intervals are already considerable and will undoubtedly expand – especially as the use of “sexed” semen becomes more routine.
On the female side, studies of normal ovulation have given researchers clues for achieving similar changes to the ovum in vitro during in-vitro maturation (IVM). IVM is an essential prelude to IVF, which, in its conventional form[6] in horses, has lagged far behind IVF in other species, including humans. Paradoxically, success with embryo production by injection of a single sperm into an egg (intracytoplasmic sperm injection, ICSI), and with cloning by stimulating the mature ovum to “re-programme” adult cells, has been at least as good in horses as in other species. Although production of horse embryos in vitro seems likely to remain an expensive laboratory procedure for the foreseeable future, research into in-vitro procedures has already had spin-offs. Thus, the development of methods of retrieving oocytes from the living mare, handling them in a proper culture medium, and transferring them (either alone or with spermatozoa) to the oviducts of young, fertile mares has produced hundreds of foals from subfertile mares and stallions.[7] Just as important is the revelation (mostly in cattle and sheep) that the environment experienced by an embryo in vitro can have effects that persist into later pregnancy, post-natally, and even into future generations. As research unravels the reasons for this, the implications for the nutrition and management of pregnant mares becomes significant indeed.
A sizeable industry has developed around the procedure of embryo transfer (ET). Argentinian polo pony breeders, for example, transferred more than 4,000 embryos at 11 centres during the 2007-2008 breeding season, allowing the elite donor mares to continue in their playing careers while recipients carried their offspring.[8] Most ET techniques have been mastered but an important exception is that the cryopreservation (“freezing”) of horse embryos, which works well on embryos recovered from the uterus about 6 days after ovulation, cannot yet be used efficiently on older embryos.
The scope of the new molecular techniques that have become available for undertaking reproductive research deserves to be underlined because the methods are being applied to all aspects of equine research. They make it possible to determine which tissues are producing what proteins and other molecules, and in what quantities, which tissues are producing receptors for those molecules, and which genes control these productions. Resultant interactions regulate the response of one tissue to another, including the response of a mare to her embryo and vice versa. The prospects for understanding the events of early pregnancy and pregnancy failure, and for developing tools for genetic selection of horses, have never been brighter.
Genetic selection in horse breeding has been practised for centuries, especially in Thoroughbreds[9], though not with the intensity and objective criteria of selection that have been used in cattle. Analysis of the chromosomes themselves (cytogenetics), however, has enabled researchers to identify important abnormalities in chromosomal structure that can cause congenital abnormalities, embryonic loss and infertility.[10]
Reproductive research in horses does not end with a firmly established pregnancy; the pregnancy has to be maintained to term and followed by successful birth and development of the foal. An example of a reproductive problem that spans early and late pregnancy is Mare Reproductive Loss Syndrome (MRLS) which caused losses estimated at $500 million to the Kentucky horse breeding industry in 2001 and 2002. Though believed to be associated with the ingestion of massive numbers of caterpillars, the exact causes of abortion in this syndrome are still under investigation.[11]
The economic impact of MRLS should remind us of the necessity to maintain scientific expertise capable of meeting unexpected disease challenges, either non-infectious (like MRLS), or infectious, like Contagious Equine Metritis (CEM) which is currently affecting the import and export of horses. | ||||
In summary, the efficiency of the horse breeding industry today depends on research of the past; its advance will depend on research of the present and future. By maintaining and expanding the symbiotic relationship between the industry and its supporting researchers, we have every prospect of keeping Canadian horse breeding abreast of the field. |
||||
|
||||
| Learn more about Dr. Keith Betteridge and the research program | ||||